"Stereochemistry of Seven-Coordinate Main Group and d0 Transition Metal Molecules". " "Non-VSEPR" Structures and Bonding in d(0) Systems". "Data-driven design of molecular nanomagnets". Cardona-Serra, Salvador Gaita-Ariño, Alejandro (). 
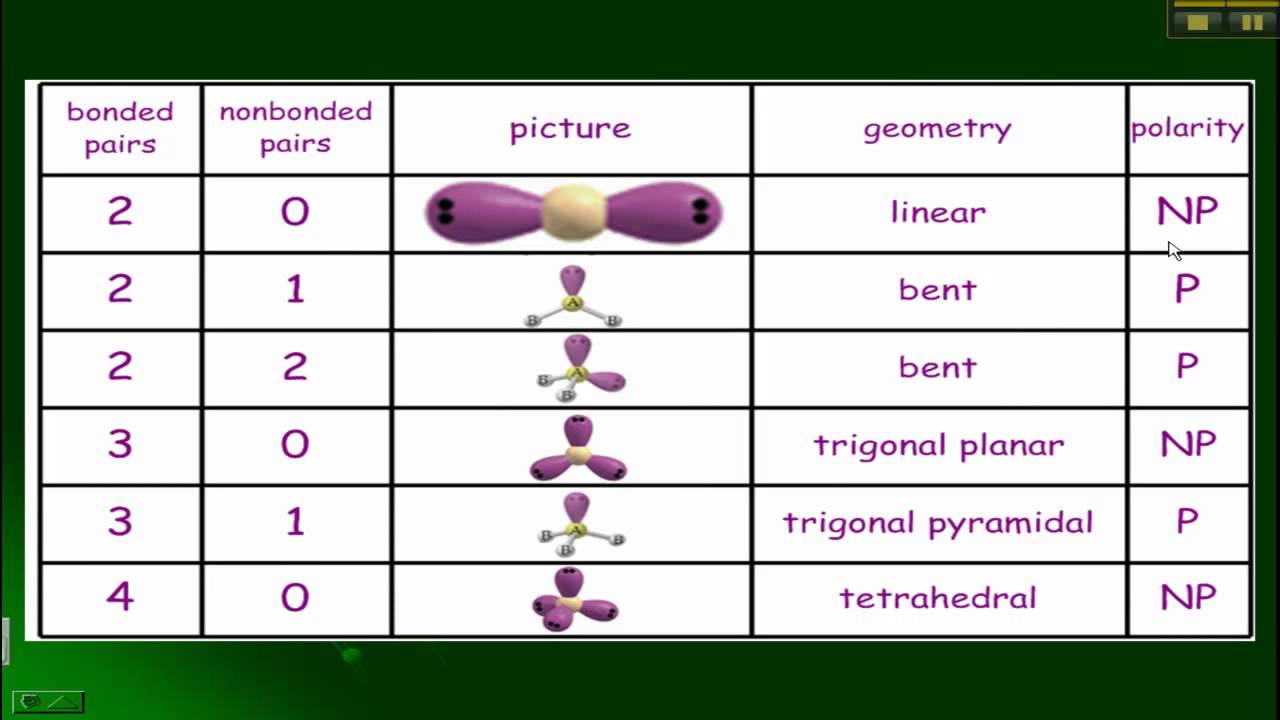
Giménez-Santamarina, Silvia Scheie, Allen Baldoví, José J. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6 Identify the electron-pair geometry based on the number of regions of electron density: linear, trigonal planar, tetrahedral, trigonal bipyramidal, or octahedral ( Figure 7.2. A molecular orbital exploration of structure, stereochemistry, and reaction dynamics". Its Lewis structure consists of double bonds between the central carbon atom and each oxygen atom. Carbon dioxide is another example of a molecule which falls under the AB2 AB 2 category. Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York: Wiley-Interscience, ISBN 7-5 The HBeH H Be H bond angle is 180o 180 o because of its linear geometry. Albert Wilkinson, Geoffrey Murillo, Carlos A. Pentagonal bipyramids are claimed to be promising coordination geometries for lanthanide-based single-molecule magnets, since (a) they present no extradiagonal crystal field terms, therefore minimising spin mixing, and (b) all of their diagonal terms are in first approximation protected from low-energy vibrations, minimising vibronic coupling. This is one of the three common shapes for heptacoordinate transition metal complexes, along with the capped octahedron and the capped trigonal prism. 
The pentagonal bipyramid is a case where bond angles surrounding an atom are not identical (see also trigonal bipyramidal molecular geometry). Fluorine belongs to the family of halogen in group 17 and has a valency of 7. Oxygen belongs to group 16, the chalcogen family, and has a valency of 6. The first and foremost step is to calculate the total number of valence electrons in an OF2 molecule. Step 1: Find the Total number of Valence Electrons. A perfect pentagonal bipyramid belongs to the molecular point group D 5h. Steps to form OF2 Lewis Structure Diagram. In chemistry, a pentagonal bipyramid is a molecular geometry with one atom at the centre with seven ligands at the corners of a pentagonal bipyramid. Structure of iodine heptafluoride, an example of a molecule with the pentagonal-bipyramidal coordination geometry. Molecular structure having atoms at the centre and corners of a pentagonal bipyramid Pentagonal bipyramidal molecular geometry
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
